GetStudySolution
Getstudysolution is an online educational platform that allows students to access quality educational services and study materials at no cost.
NCERT Solutions for Class 9 Science chapter 4 – Structure of the Atom
Back Exercise
Question 1.
Compare the properties of electrons, protons and neutrons.
Answer:

Question 2.
What are the limitations of J.J. Thomson’s model of an atom ?
Answer:
Limitations of Thomson’s model of an atom
(i) It could not explain the result of the scattering experiment performed by Rutherford.
(ii) It did not have any experimental evidence in its support.
Question 3.
What are the limitations of Rutherford’s model of an atom ?
Answer:
Limitations of Rutherford Model atom
- Rutherford model of atom could not explain the stability of the atom.
- Rutherford model of atom could not explain as to how the electrons are distributed in the extra nuclear portion in an atom.
Question 4.
Describe Bohr’s model of an atom.
Answer:
In 1913, Neils Bohr gave a theory regarding the distribution of electrons in the extra nuclear space in an atom. The main postulates of the theory are listed :
- In the extra nuclear portion of an atom, the electrons revolve in well defined circular paths known as orbits.
- These circular orbits are also known as energy levels or energy shells.
- These have been designated as K, L, M, N, O, … (or as 1, 2, 3, 4, 5, …) based on the energy present.
- The order of the energy of these energy shells is :
K<L<M<N<0 <…. or 1< 2< 3 < 4<5 <…. - While revolving in an orbit, the electron is not in a position to either lose or gain energy. In other words, its energy remains stationary. Therefore, these energy states for the electrons are also known as stationary states.
Question 5.
Compare all the proposed models of an atom given in this chapter.
Answer:
For Thomson’s Model of air atom:
- Sphere is positively charged
- Electrons are negatively charged and scattered all through the inside of the sphere.
- Positively charged = negatively charged
- The net charge in the atom is zero.
For Rutherford’s Model of an atom:
- The nucleus is at the centre and is positively charged holding the entire mass.
- Electrons are negatively charged revolving in a well-defined path
- In comparison with the nucleus, the size of the atom is very large.
For Bohr’s Model of air atom:
- Nucleus is present at the centre and is positively charged
- Electrons are negatively charged, revolving around but do not radiate energy.
- The distinct orbits are labelled as K, L, M, N
Question 6.
Summarise the rules for writing the distribution of electrons in various shells for first eighteen elements.
Answer:
As the electrons have negligible mass, the entire mass of the atom is that of protons and neutrons present in its nucleus. Since each proton and neutron has mass equal to 4 u, therefore, mass of an atom must be the same as the sum of protons and neutrons present in the nucleus. The latter is known as mass number of an element. It is denoted by the symbol A’. The mass number of an element may be defined as :
The sum of the number of protons and neutrons present in the nucleus of its atom.
Mass number (A) = No. of protons + No. of neutrons
Question 7.
Describe valency by taking the examples of silicon and oxygen.
Answer:
Valency of an element may be defined as the combining capacity of its atom with atoms of other elements in order to acquire 8 electrons (2 in some exceptional cases).
Valency of silicon (Si) : Atomic number of the element is 14. Its electronic distribution is ; K(2), L(8), M(4).
As silicon atom has four valence electrons, it can lose all of them. At the sametime, it can also gain four electrons to have a complete octet. Therefore, the valency of silicon is 4.
Valency of oxygen (O) : Atomic number of the element is 8. Its electronic distribution is ; K(2), L(6)
As oxygen atom has six valence electrons, it needs two more electrons to complete its octet (8-6 = 2). Therefore, valency of oxygen is 2.
Question 8.
Explain with examples
(i) Atomic number
(ii) Mass number
(iii) Isotopes
(iv) Isobars.
Answer:
(i) Atomic number:
Rutherford stated that the number of unit positive charges present in the nucleus of an atom is known as atomic number of the element It is denoted by the symbol Z.
Atomic no. (Z)= No. ofprotons = No. of electrons
(ii) Mass number:
The mass number of an element may be defined as : the sum of the number of protons and neutrons present in the nucleus of its atom.
Mass number (A) = No. of protons + No. of neutrons
(iii) Isotopes:
Isotopes may be defined as the different atoms of the same element having same atomic number but different mass numbers.(iv) Isobars:
isobars may be defined as the atoms belonging to the different elements with same mass numbers but different atomic numbers.
Question 9.
Na+ ion has completely filled K and L shells. Explain. (CBSE 2013)
Answer:
Na+ ion is formed when Na atom loses one electron from the valence shell i.e. M shell. The Na+ ion formed has only two shells which are complete
Na : K(2) L(8) M(1) ;
Na+ : K(2) L(8)
Question 10.
If the element bromine is in the form two isotopes which are 7935Br (49.7%) and 8135Br (30.3%), then calculate the average atomic mass of bromine.
Answer:
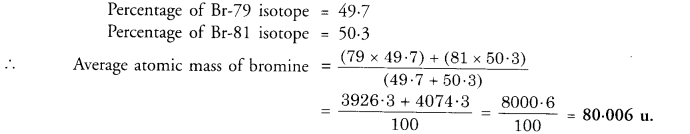
Question 11.
The average atomic mass of a sample of element X is 16.2 u. What are the percentages of isotopes 168X and 188X in the sample ? (CBSE 2016)
Answer:
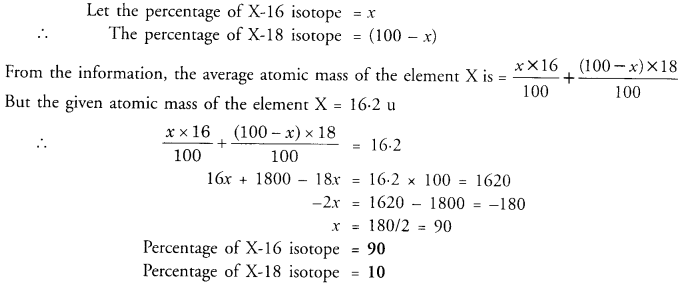
Question 12.
If Z = 3, what would be the valency of the element ? Also name the element.
Answer:
The configuration of the element with Z = 3 is K(2) L(l). It has one electron in the valence shell i.e. L-shell. Its valency is one. The element is lithium (Li).
Question13.
Composition of the nuclei of two atomic species X and Y are given as under
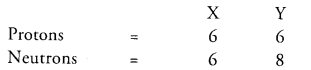
Give the mass numbers of X and Y. What is the relation between the two species ?
Answer:
Mass number of X = protons + neutrons = 6 + 6 =12 Mass number of Y = 6 + 8 = 14
The species X and Y are the isotopes because their atomic numbers are same and their mass numbers are different.
Question 14.
For the following statements, write T for True and F for False.
(a) J.J. Thomson proposed that the nucleus of an atom contains only nucleons.
(b) A neutron is formed by an electron and proton combining together. Therefore, it is neutral.
(c) The mass of an electron is about 1/2000 times that of proton.
(d) An isotope of iodine is used for making tincture of iodine, which is used as medicine.
Answer:
(a) F
(b) F
(c) T
(d) F.
Question 15.
Rutherford’s alpha-particle scattering experiment was responsible for the discovery of
(a) Atomic Nucleus
(b) Electron
(c) Proton
(d) Neutron.
Answer:
(a) is the correct answer.
Question 16.
Isotopes of element have :
(a) the same physical properties
(b) different chemical properties
(c) different number of neutrons
(d) different atomic numbers.
Answer:
(c) Different number of neutrons.
Question 17.
Number of valence electrons in Cl– ion are :
(a) 16
(b) 8
(c) 17
(d) 18.
Answer:
(b) 8
Question 18.
Which one of the following is a correct electronic configuration of sodium ?
(a) 2, 8
(b) 8, 2, 1
(c) 2, 1, 8
(d) 2, 8, 1
Answer:
(d) is the correct answer.
Question 19.
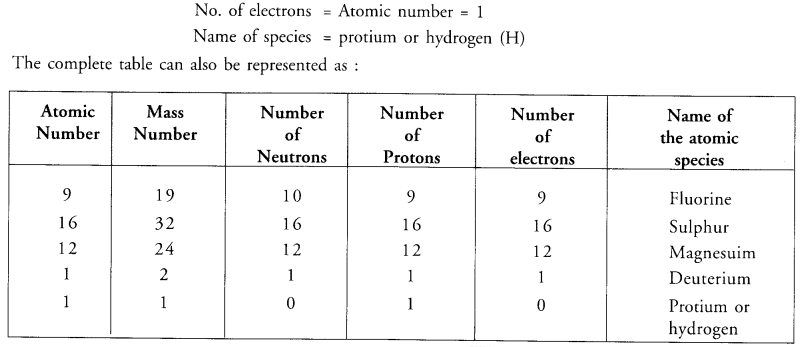
Complete the following table.
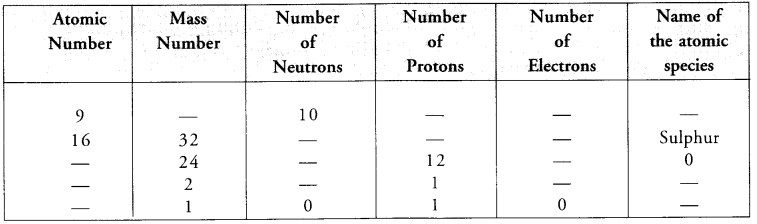
Answer:

In-Text Questions
Question 1.
What are canal rays ?
Answer:
Canal rays also called anode rays, are seen moving from the anode towards the cathode in the specially designed discharge tube. However, they do not originate from the anode.
Question 2.
If an atom contains one electron and one proton, will it carry any charge or not ?
Answer:
No, it will not carry any charge because an electron has a unit negative charge and a proton has a unit
positive charge. They neutralise each other.
Question 3.
On the basis of Thomson’s model of an atom explain how the atom is neutral as a whole.
Answer:
According to Thomson’s Model of an atom, an atom may be regarded as a positively charged sphere in
which protons are present. The negatively charged electrons are believed to be studded or embedded in the sphere. Since the negative charges due to electrons and positive charges due to protons balance each other, the atom as a whole is neutral.
Question 4.
On the basis of Rutherford’s model of an atom, which sub-atomic particle is present in the nucleus of an atom ?
Answer:
The nucleus of atom is positively charged according to Rutherford’s model of an atom. All the protons in an atom are therefore, present in the nucleus.
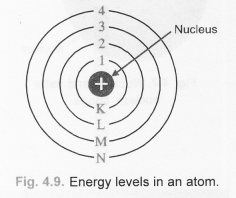
Question 5.
Draw a sketch of Bohr’s model of an atom with three shells.
Answer:
Question 6.
What do you think would be the observation if the a-particle scattering experiment is carried out using a foil of a metal other than gold ?
Answer:
If the a-scattering experiment is performed using a foil of a metal rather than gold, there would be no change in the observation. In the a-scattering experiment, a gold foil was taken because gold is a malleable and a thin foil of gold can be easily made than foils of other metals.
Question 7.
Name the three sub – atomic particles in an atom.
Answer:
The sub-atomic particles in an atom are : protons, electrons and neutrons.
Question 8.
Helium atom has atomic mass of 4u and has two protons in its nucleus. How many neutrons does it have ?
Answer:
Mass number of helium is equal to its atomic mass but has no units.
∴ Mass number (A) of helium = 4
No. of protons in the nucleus = 2 Atomic number (Z) of the element = 2
No. of neutrons (n) = A – Z = 4 – 2 = 2.
Question 9.
Write the distribution of electrons in carbon and sodium atoms.
Answer:
Carbon(Z = 6) : 2 (K-shell) ; 4(L-shell)
Sodium (Z = 11) : 2 (K-shell) ; 10 (L-shell) ; 1 (M-shell)
Question 10.
If the K and L shells of an atom are full, then what would be the number of electrons in the atom ?
(CBSE 2014)
Answer:
Maximum no. of electrons in K-shell = 2
Maximum no. of electrons in L-shell = 8
Total no. of electrons in the atom = 2 + 8 = 10
Question 11.
If the number of electrons in an atom is 8 and the number of protons is also 8, then :
(i) What would be the atomic number of the atom ?
(ii) What is the charge on the atom ?
Answer:
(i) Atomic number (Z) of the atom = No. of protons = No. of electrons = 8
(ii) Charge on the atom = No charge
Actually, when the number of protons and electrons are the same, the atom is neutral.
Question 12.
For the symbols H, D and T, tabulate three sub-atomic particles found in each of them.
Answer:

Question 13.
Write the electronic configuration of any pair of isotopes and isobars.
Answer:
(i) Electronic configuration of pair of isotopes :

(ii) Electronic configuration of pair of isobars :
Isobars have same mass number but different atomic numbers. For example, elements calcium and argon are isobars :
Ca(Z = 20) : 2, 8, 8, 2
Ar (Z = 18) : 2, 8, 8.
Please note that both the elements have mass number (A) equal to 40.